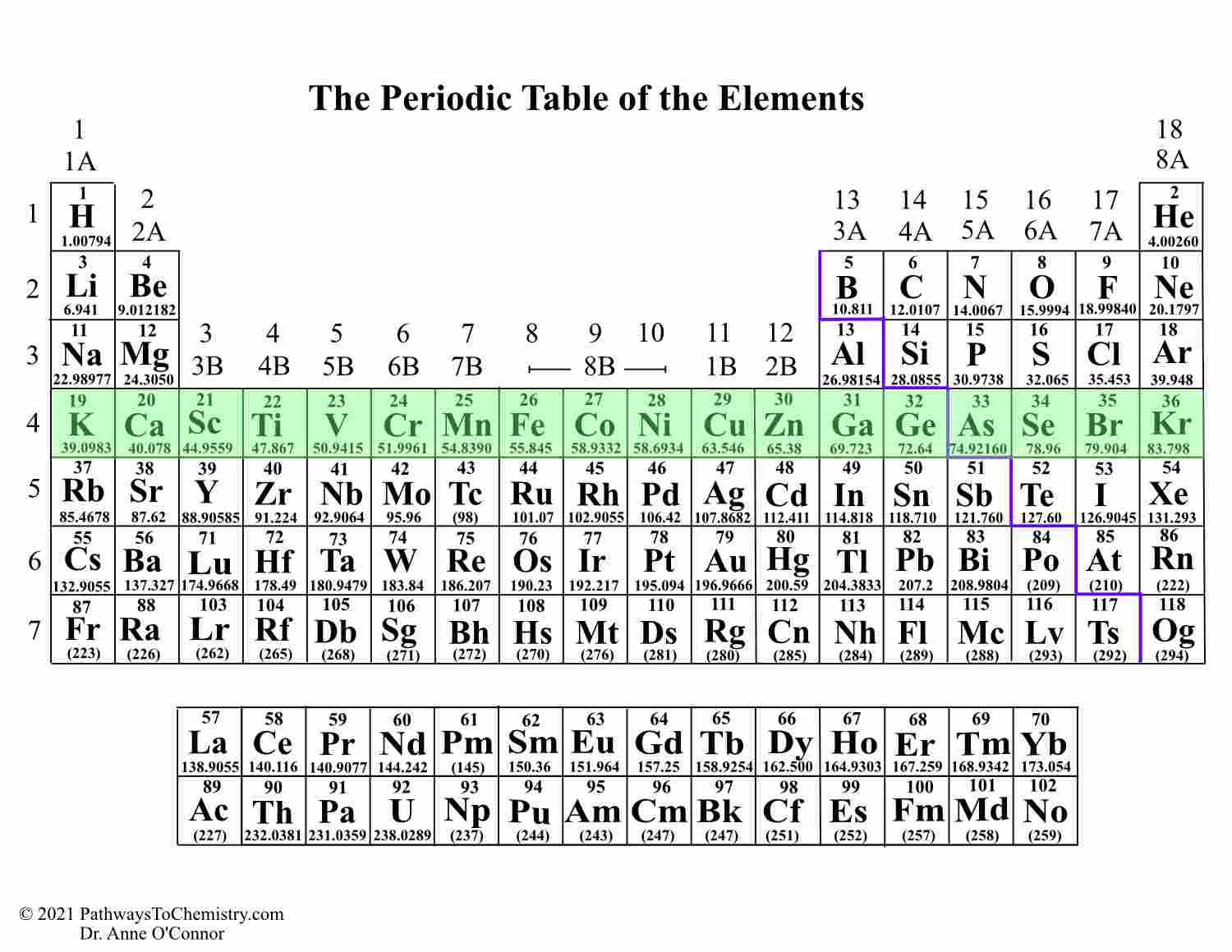
You can effortlessly find every single detail about the elements from this single Interactive Periodic table. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). They get more reactive the further down on the periodic table you go too, with cesium and francium being so reactive that they can burst into flames simply by being exposed to the air. Simple Chemical Reactions Unit Test 3 of 193 of 19 Items Question Use the periodic table to answer the question. Alkali metals react vigorously to water and air.These elements will dance around, sizzling due to the production of hydrogen gas, and often explode. Free Gift for you: Interactive Periodic Table Top answer: The two groups of elements in the periodic table that are the most reactive are the alkali metals in Read more. Fluorine is identified as the most reactive nonmetal and the most electronegative element in the periodic table, making it the strongest oxidizing agent. Understand the patterns in formula of compounds. In this way, the elements of the same group show similar chemical properties and they also have the same number of valence electrons. Fluorine is a highly reactive element, and form fluorides with the majority of other elements. Most of them react with atmospheric oxygen to form metal oxides. Metals tend to readily lose electrons and form cations. Other chemical properties include: Electropositive Character: Metals tend to have low ionization energies, and typically lose electrons (i.e. Note: Astatine (atomic number 85) shows characteristics of nonmetals (halogens) as well as metalloids. 3,093 A chart of the reactivity series of common metals is provided below. All these nonmetals are located on the upper right corner of the Periodic table ( Hydrogen is located on the left top corner) In the above image, the nonmetals are represented in yellow color. They are soft and can be cut easily with a kitchen knife.Īlso all the elements of group 1 have one valence electron.Īll the elements of group 18 are chemically inert (that means they do not easily react with other elements).Īnd all the elements of group 18 have a complete octet (that means they have 8 electrons in their outer shell). There are 18 nonmetals on the Periodic table. 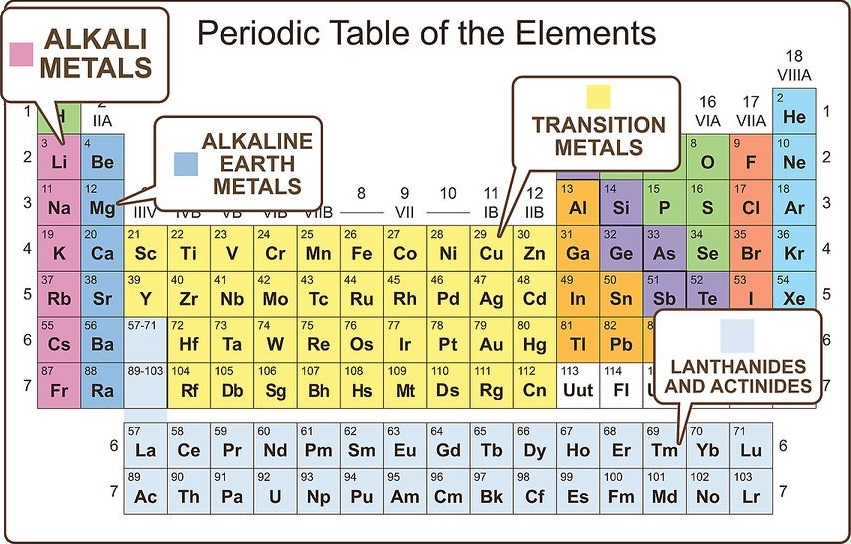
The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons.Īll the elements of group 1 are highly reactive to water. There are total 18 vertical columns on periodic table. Group 3-12: Transition and Inner transition metals group. Group 1: Alkali metals group (hydrogen not included) Group 2: Alkaline earth metals group. Groups are the vertical columns on the periodic table. There are total 18 different groups in Periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
